Development of validated methods for QC testing
For many SMEs, entering regulated supply chains – whether in defence, pharmaceuticals, or advanced manufacturing – is a major milestone. But it comes with a critical requirement: robust, validated analytical methods that can stand up to audit and scrutiny.
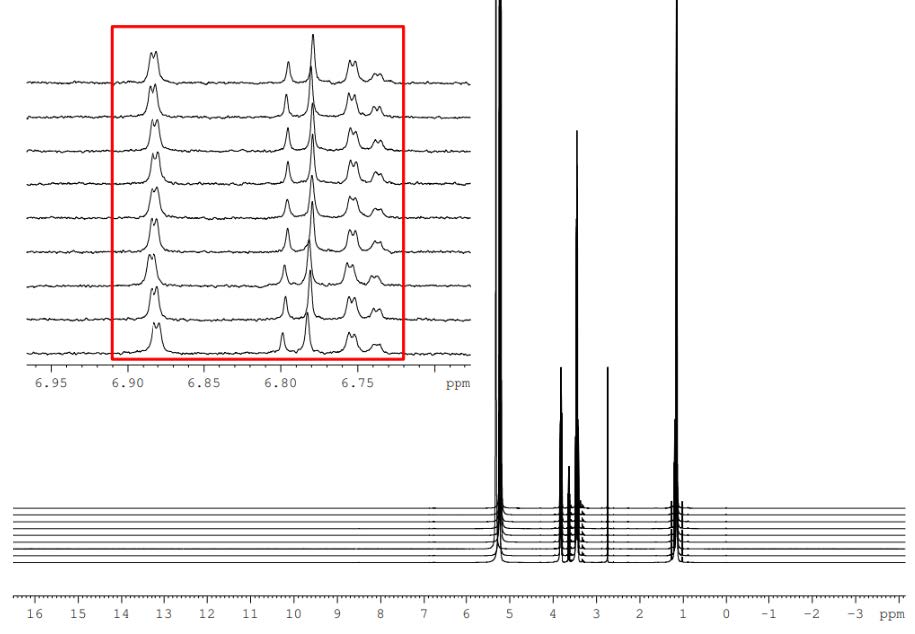
At The Bridge, we recently supported a Lincolnshire SME in developing a fully validated QC method to meet stringent Government contract requirements. By combining advanced instrumentation with GxP-aligned processes, we delivered a reliable, reproducible, and audit-ready solution.
- Enabled compliance with strict tolerances
- Delivered defensible, high-integrity data
- Created a transferable, long-term QC asset
- Removed the need for costly in-house capability
This project highlights the value of collaboration to bring together academic expertise and industrial rigour to solve real-world challenges Read the full case study below to see how validated methods can accelerate your route into high-value markets.
Download Exemplar Study